|
These range from liquids to hard, glasslike solids with many useful properties.Įlemental silicon transmits more than 95% of all wavelengths of infrared and and has been used in lasers to produce coherent light at 456 nm. Hydrolysis and condensation of substituted chlorosilanes can be used to produce a very great number of polymeric products, or silicones. Workers in environments where silicaceous dust is breathed may develop a serious lung disease known as silicosis. Silicon shares the bonding versatility of carbon, with its four. It is a group 14 element in the same periodic group as carbon, but chemically behaves distinctly from all of its group counterparts. While atomic mass is an absolute mass, relative isotopic mass is a dimensionless number with no units. Hence the silicon element has electrons arrangement 2, 8, 4. Relative isotopic mass (a property of a single atom) is not to be confused with the averaged quantity atomic weight (see above), that is an average of values for many atoms in a given sample of a chemical element. One of its forms is shiny, grey, and very brittle (it will shatter when struck with a hammer). Now the atomic number of silicon (Si) is 14. Silicon carbide is one of the most important abrasives. Silicon is a crystalline semi-metal or metalloid. Silicon is an important ingredient in steel. Diatoms in both fresh and salt water extract silica from the water to use as a component of their cell walls. Silicon is important in plant and animal life. In a typical sample of carbon-containing material, 98.89 of the carbon atoms also contain 6 neutrons, so each has a mass number of 12. The element carbon (C) has an atomic number of 6, which means that all neutral carbon atoms contain 6 protons and 6 electrons. Silicon is found also in minerals such as asbestos, feldspar, clay and mica. Mass Number(A) Number of Protons + Number of Neutrons. It is found largely as silicon oxides such as sand (silica), quartz, rock crystal, amethyst, agate, flint, jasper and opal. 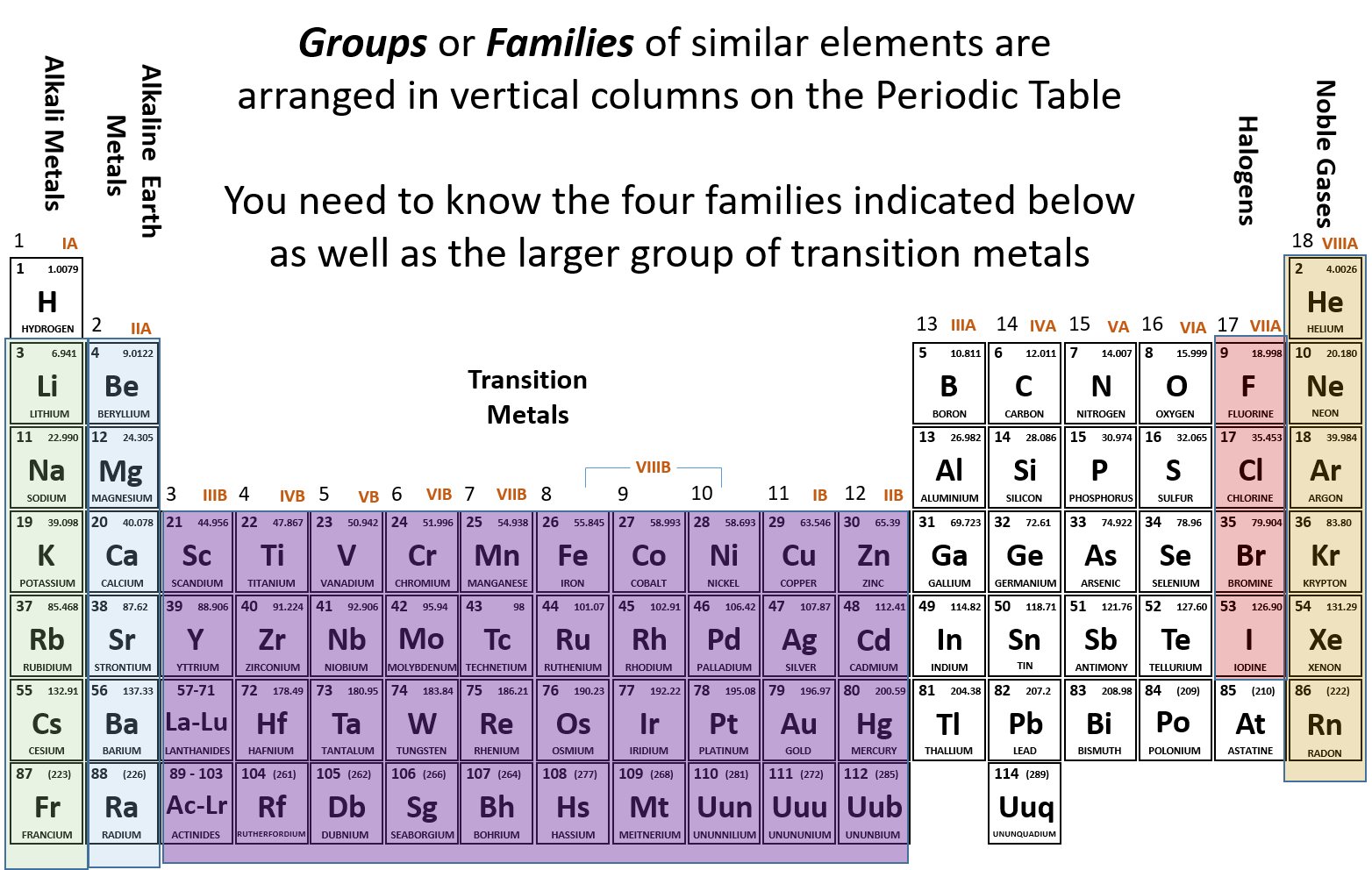
Silicon makes up 25.7% of the earth's crust by weight, and is the second most abundant element, exceeded only by oxygen. Silicon is present in the sun and stars and is a principal component of a class of meteorites known as aerolites.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
